Search
Sharing Knowledge improves Knowledge... Knowledge should come at as less cost as possible.
Posts
Showing posts from February, 2013
Posted by
Varun C N
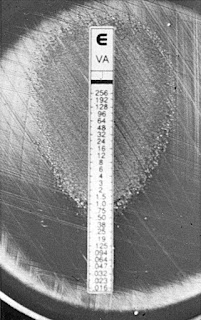
Some bacteria loves antibiotic
- Get link
- Other Apps

Posted by
Varun C N
Say Hi to "Methanogens in human flora"
- Get link
- Other Apps

Posted by
Varun C N
Schwan cell programmer- Leprae
- Get link
- Other Apps