Search
Sharing Knowledge improves Knowledge... Knowledge should come at as less cost as possible.
Posts
Showing posts from 2015

Posted by
Varun C N
Break Announcement- 2
- Get link
- X
- Other Apps

Posted by
Varun C N
Antibiotic Catastrophe- MCR1: Follow up
- Get link
- X
- Other Apps

Posted by
Varun C N
The Red and Black Queen- Hypothesis
- Get link
- X
- Other Apps

Posted by
Varun C N
BtB#5- Defining Immune privilege
- Get link
- X
- Other Apps

Posted by
Varun C N
Global Antibiotic Resistance Alert: MCR-1 in Plasmid
- Get link
- X
- Other Apps
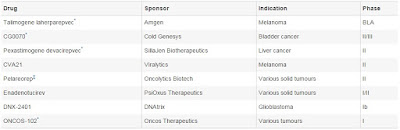
Posted by
Varun C N
Dead bacteria fights Lab model of Colorectal cancer
- Get link
- X
- Other Apps

Posted by
Varun C N
Toxoplasma Vaccine: A step Closer
- Get link
- X
- Other Apps

Posted by
Varun C N
Lab series# 8: PBMC Isolation technique
- Get link
- X
- Other Apps

Posted by
Varun C N
BtB#5- An intro to Bacteriophages
- Get link
- X
- Other Apps

Posted by
Varun C N
Hitting the RNA switch: A new method of antibiotic design
- Get link
- X
- Other Apps

Posted by
Varun C N
Nobel Awards- 2015
- Get link
- X
- Other Apps