Will go to war for Iron: Bacterial strategy
Greetings
There is a great deal of interest in studying what contributes to pathogenesis and virulence. I have recently posted on "Host-parasite interaction", and mentioned about "Nutritional immunity". A lot of literature on Nutritional immunity is based on the competition for iron. Iron forms an important part of a lot of enzymes in cellular machinery. The requirement for iron is of paramount importance for host and parasite. Most of the bacteria studied have a chunk of genome dedicated to acquiring and storage of iron.
Recently there was a discussion in TWiM 111 (Link), regarding targeting iron acquisition system as a possible antibiotic. The discussion was based on a proof of concept paper showing that gallium which is a close mimic of iron, can be a potential attack molecule. Of course gallium has significant toxicity issues and has limited use when it comes to clinical application. However, this paper raises the idea that inhibiting iron acquisition in bacteria is a potential antibiotic. Though the paper describes that this will be a very difficult antibiotic to which resistance can be acquired, it is difficult to consider that pragmatically it will hold.
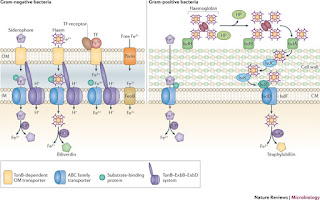 |
| Fig 1: Example of Iron acquisition system. Source |
It is a common assumption that iron is a very special nutrient and hence ever accumulated (whenever available). This is not true. Iron in exceeding quantities can have toxic effects. There are regulatory systems in bacteria which serves to shut down the acquisition system when iron has reached high levels. However, questions have been raised on, if ever a bacteria reaches such a state considering the fierce competition for iron. Studies on these systems have been of great interest in past few years. As Professor Peter Williams comments, "There has been an explosion in research on iron metabolism in pathogenic bacteria. A deeper knowledge of the mechanisms involved in iron scavenging could result in medically useful advances". Proposal of attenuating bacteria for vaccines by removing the iron regulatory systems, antibiotics designed to target these systems and attaching antibiotic to siderophores etc are a couple of examples.
Interestingly, it appears that iron acquisition system is also linked to quorum sensing molecules. Recently, this has been explored in a paper by Nguyen etal. They studied the effect of iron depletion in terms of polymicrobial interactions. It is known that P aeruginosa can secrete 2-alkyl-4(1H)-quinolones (shortly known as AQ) leading to lysis of S. aureus. In a state of low iron content, the AQ mediated lysis helps in recovery of iron storage from S aureus. It is quite possible that because of the iron limitation factor, polymicrobial infections may be much less fulminant than assumed to be, at least in Cystic fibrosis case (with Pseudomonas as one of the infecting agents) as the paper suggests.
The study has several implications. But one of them that had me digging (not discussed in the paper) was how is this going to play out if we are thinking of using engineered bacteria or predatory bacteria as antibiotic alternatives.
The study has several implications. But one of them that had me digging (not discussed in the paper) was how is this going to play out if we are thinking of using engineered bacteria or predatory bacteria as antibiotic alternatives.
Ross-Gillespie A, Weigert M, Brown SP, & Kümmerli R (2014). Gallium-mediated siderophore quenching as an evolutionarily robust antibacterial treatment. Evolution, medicine, and public health, 2014 (1), 18-29 PMID: 24480613
Skaar EP (2010). The battle for iron between bacterial pathogens and their vertebrate hosts. PLoS pathogens, 6 (8) PMID: 20711357
Nguyen AT, Jones JW, Ruge MA, Kane MA, & Oglesby-Sherrouse AG (2015). Iron Depletion Enhances Production of Antimicrobials by Pseudomonas aeruginosa. Journal of bacteriology, 197 (14), 2265-75 PMID: 25917911





Comments
Post a Comment