New interest in Hecolin
Hello,
First, i must agree that i have been busy working on various aspects and just haven't got sufficient time to post microbiology. But then, i have lots to say. I have been thinking if i could post more than once a week just to cover up what i want to say. But then, time makes a factor. Learning new exciting things takes time and i believe you agree. Also I'm glad to see that my blog rank has significantly increased as per the technorati ranking. That motivates me to write more anyway.
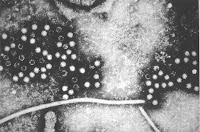 |
| Photo 1: Hepatitis E virus (Source) |
I have so many stories to talk about. However, i have been intending to write on this or long. Hepatitis E virus (HEV), is an enterically-transmitted acute viral hepatitis. The infection was first documented in 1955, is a member of the genus hepevirus in the family Hepeviridae. This is a an icosahedral, non-enveloped with single stranded positive-sense RNA genome. This virus has been sequenced and there appears to be at least 4 genotypes. Genotype 1 and 2 are human specific, but genotype 3 and 4 infect humans and animals. Genotype 3 has been linked to least virulence. Read more here. The infections have an incubation period of nearly 6 weeks (2- 9 weeks), and becomes clinically apparent in about 10% of the cases. The infection is usually self limiting, but has devastating effects in pregnant women (Reference).
There is no FDA licensed vaccine available for HEV. Hecolin was a prototype vaccine that had successfully entered clinical trials. It showed promising early results. The contents of vaccine was called as HEV 239 (30 microg of purified recombinant hepatitis E antigen adsorbed to 0.8 mg aluminium hydroxide suspended in 0.5 mL buffered saline) (Reference). This has happened in 2010.

 |
| Photo 2: Hecolin Vaccine (Source) |
So, why am i pulling this out now? Its because of renewed interest, since the vaccine has been approved in China in 2011 by SFDA (China’s State Food and Drug Administration). Hecolin will be marketed by Xiamen Innovax Biotech Co. Ltd. Zhang, an investigator in the process says “Many people — including representatives of multinational pharmaceutical companies, venture capitalists, Chinese local government officials and Chinese entrepreneurs — think this is a worthy example of biotechnology investment,” he says". (Source).
I wouldn't be surprised if the WHO decides to join hands with the Innovax and start vaccinating people around the world.
Zhu FC, Zhang J, Zhang XF, Zhou C, Wang ZZ, Huang SJ, Wang H, Yang CL, Jiang HM, Cai JP, Wang YJ, Ai X, Hu YM, Tang Q, Yao X, Yan Q, Xian YL, Wu T, Li YM, Miao J, Ng MH, Shih JW, & Xia NS (2010). Efficacy and safety of a recombinant hepatitis E vaccine in healthy adults: a large-scale, randomised, double-blind placebo-controlled, phase 3 trial. Lancet, 376 (9744), 895-902 PMID: 20728932
Park, S. (2012). Hepatitis E vaccine debuts Nature, 491 (7422), 21-22 DOI: 10.1038/491021a





Comments
Post a Comment