Attacking HIV Rebounce
Greetings
HIV is an infection of great interest to international community. Despite years of research, neither has there been a successful vaccine nor a complete treatment. Needless to say we have been able to better the treatment options than it was a decade before. Most of the infected individuals have been able to live an extended normal life thanks to some of the best anti- retroviral drugs. The current research is aimed more at finding a complete cure rather than just finding new drugs. Pinnacle would be a successful vaccine. Recently an article was published in Nature news (Link) arguing that current HIV pandemic could be handled in a better way using current existing tools.
As I have explained several times in my earlier posts, the grave problem in flushing the HIV out completely is the lack of ability to flush the latent virus. Studies have tried to tackle the issue by a variety of approaches such as DNA editing techniques, HDAC inhibitors etc. Though these approaches have promised potentials, its usefulness in treatment has been questioned. The key player in regulation of latency is the TAT molecule. I have argued in an older post (Link) that TAT is a good candidate to attack. I mentioned didehydro- Cortistatin A as a potential molecule.
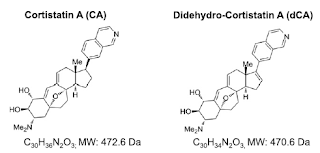 |
| Fig 1: Structure of CA and its analog dCA. Source |
 |
| Fig 2: dCA mediated TAT inhibition. Source |
So you see, TAT is a very attractive candidate for attack. It is virus specific, no similar protein in host tissue, early viral expressed protein and key regulator of HIV latency. Earlier studies have characterized didehydro- Cortistatin A (A derivative of naturally occurring steroidal alkaloid Cortistatin A also referred as dCA) can inhibit the TAT feedback system thus effectively inhibiting the viral expression. The studies showed that dCA suppresses viral particle release from CD4+T cells. In other words they impair the ability to reactivate from latency.
A more recent study published by Mousseau et al; has looked into more details of this mechanism and showed that use of dCA along with ART (Anti Retroviral Therapy) may provide a excellent way of reducing viral rebounce. There are of course a couple of issues. For example, this is not an absolute therapy. There is a level of basal transcription of virus that can happen in the absence of TAT. This can be seen from the data in the study showing that when tested against patient cells, dCA reduced an average of 92.3% of the viral reactivation in lab stimulating conditions.
Now here's the punchline. When dCA was withdrawn, the block still appears to work indicative of a permanent change or at least a long-term change. The authors suggested that the promoter is probably altered. Based on these findings they hypothesized two possibilities (Quote from paper) (i) dCA simply accelerates the establishment of typical proviral latency by facilitating the corresponding epigenetic modifications; (ii) because dCA blocks a subset of the multiple Tat activities, it may result in unique epigenetic changes.
A yet another mechanism that is less discussed is targeting TAT degradation. If a compound can specifically increase the proteosomal degradation rate for TAT, it could be equally effective in preventing reactivation. One such compound is Triptolide. Currently this is another possible drug under investigation. Research has also been done one antibodies that may target TAT domain directly, However not much convincing data is available in this case. I have also heard of designs of TAR decoys and possible chemicals that may attack the TAT-TAR interaction. However, I have not seen a convincing data on these too. Perhaps readers can suggest me of any paper they known of.
To conclude and make a statement, TAT is one of the leading molecules preferred to be attacked. With molecules being identified that has potential activity. Pharma companies are bound to pick these and make modifications to yield better chemicals, that may ultimately help us in significant betterment of HIV therapy.
Now here's the punchline. When dCA was withdrawn, the block still appears to work indicative of a permanent change or at least a long-term change. The authors suggested that the promoter is probably altered. Based on these findings they hypothesized two possibilities (Quote from paper) (i) dCA simply accelerates the establishment of typical proviral latency by facilitating the corresponding epigenetic modifications; (ii) because dCA blocks a subset of the multiple Tat activities, it may result in unique epigenetic changes.
 |
| Fig 3: Structure of Tritolide. Source |
To conclude and make a statement, TAT is one of the leading molecules preferred to be attacked. With molecules being identified that has potential activity. Pharma companies are bound to pick these and make modifications to yield better chemicals, that may ultimately help us in significant betterment of HIV therapy.
Mousseau G, Clementz MA, Bakeman WN, Nagarsheth N, Cameron M, Shi J, Baran P, Fromentin R, Chomont N, & Valente ST (2012). An analog of the natural steroidal alkaloid cortistatin A potently suppresses Tat-dependent HIV transcription. Cell host & microbe, 12 (1), 97-108 PMID: 22817991
Mousseau G, Kessing CF, Fromentin R, Trautmann L, Chomont N, & Valente ST (2015). The Tat Inhibitor Didehydro-Cortistatin A Prevents HIV-1 Reactivation from Latency. mBio, 6 (4) PMID: 26152583
Wan Z, & Chen X (2014). Triptolide inhibits human immunodeficiency virus type 1 replication by promoting proteasomal degradation of Tat protein. Retrovirology, 11 PMID: 25323821




Comments
Post a Comment